 
Uses thus far have been limited because of its high cost. Full electron configuration of krypton: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6. Krypton is used in certain photographic flash lamps for high-speed photography. 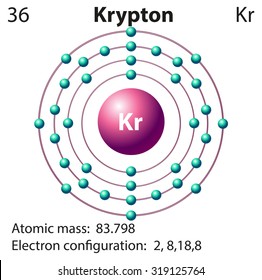
Concept: P - Block Group 18 Elements - Concept of Group. To obtain a +1 charge it loses an electron, resulting in a configuration of Ar. Electronic configuration of krypton (Z 36) is, 36Kr - 1s22s22p63s23p63d104s24p6 or Ar3d104s24p6. Estimates of the concentration of reactants are therefore made possible. Potassium (K) is orignially in the electron configuration of Ar4s1. The activity of these kryptonates is sensitive to chemical reactions at the surface. By imbedding the isotope in various solids, kryptonates are formed. 85Kr has found recent application in chemical analysis. Krypton clathrates have been prepared with hydroquinone and phenol. Molecule-ions of ArKr+ and KrH+ have been identified and investigated, and evidence is provided for the formation of KrXe or KrXe+. A higher fluoride of krypton and a salt of an oxyacid of krypton also have been reported. Krypton difluoride has been prepared in gram quantities and can be made by several methods. While krypton is generally thought of as a rare gas that normally does not combine with other elements to form compounds, it now appears that the existence of some krypton compounds is established. The spectral lines of krypton are easily produced and some are very sharp. Seventeen other unstable isotopes are now recognized. Naturally occurring krypton contains six stable isotopes. It is characterized by its brilliant green and orange spectral lines. Solid krypton is a white crystalline substance with a face-centered cubic structure which is common to all the "rare gases." Properties The atmosphere of Mars has been found to contain 0.3 ppm of krypton. Krypton is present in the air to the extent of about 1 ppm. 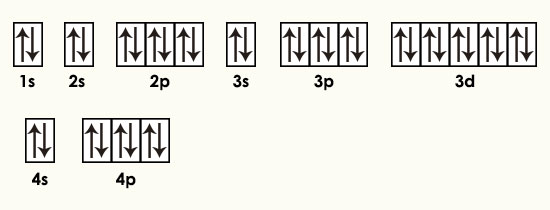
In October 1983 the meter, which originally was defined as being one ten millionth of a quadrant of the earth's polar circumference, was again redefined by the International Bureau of Weights and Measures as being the length of a path traveled by light in a vacuum during a time interval of 1/299,792,458 of a second. This replaced the standard meter of Paris, which was defined in terms of a bar made of a platinum-iridium alloy. In 1960 it was internationally agreed that the fundamental unit of length, the meter, should be defined in terms of the orange-red spectral line of 86Kr. For example, the configuration of magnesium could be written Ne3s 2, rather than writing out 1s 2 2s 2 2p 6 3s 2. The common shorthand notation is to refer to the noble gas core, rather than write out the entire configuration. kryptos, hidden) Discovered in 1898 by Ramsay and Travers in the residue left after liquid air had nearly boiled away. Krypton electron configuration Electronic configurations of elements Kr (Krypton) is an element with position number 36in the periodic table. The p sublevel can hold six electrons, the d sublevel can hold 10 electrons, and the f sublevel can hold 14 electrons. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
